Why Amylyx is pulling ALS drug Relyvrio from US market after study
WASHINGTON (AP) — The maker of a drug for Lou Gehrig’s disease that recently failed in a large study said Thursday it will pull the medicine from the market, acknowledging it didn’t help patients with the deadly neurological condition.
Amylyx Pharmaceuticals announced it will voluntarily halt sales and marketing of the drug in the U.S. and Canada, where new patients will no longer be able to get a prescription.
“While this is a difficult moment for the ALS community, we reached this path forward in partnership with the stakeholders who will be impacted and in line with our steadfast commitment to people living with ALS,” company co-founders said in a statement. Patients already taking the therapy who wish to continue will be able to enroll in a program to receive it for free.
The Food and Drug Administration approved the much-debated drug, Relyvrio, in September 2022, following a years-long advocacy campaign by patients with amyotrophic lateral sclerosis, or ALS.
Related articles

'The Apprentice,' about a young Donald Trump, premieres in Cannes
CANNES, France (AP) — While Donald Trump’s hush money trial entered its sixth week in New York, an o2024-05-21Eye watering amount of money for south Auckland roading project, cyclists say
Photo: Supplied / Google Maps2024-05-21Government thinks disabled 'add no value to society'
People who attended a rally at the Bridge of Remembrance in Christchurch on Thursday. Photo: RNZ / N2024-05-21Alec Baldwin pleads not guilty to Rust shooting charge
Actor Alec Baldwin. Photo: AFP / ANGELA WEISS2024-05-21
Pentagon vows to keep weapons moving to Ukraine as Kyiv faces a renewed assault by Russia
WASHINGTON (AP) — Defense Secretary Lloyd Austin committed Monday to keeping U.S. weapons moving to2024-05-21King Charles leaves hospital as Kate recovers at home
King Charles leaves The London Clinic with wife, Queen Camilla. Photo: AFP / ADRIAN DENNIS2024-05-21


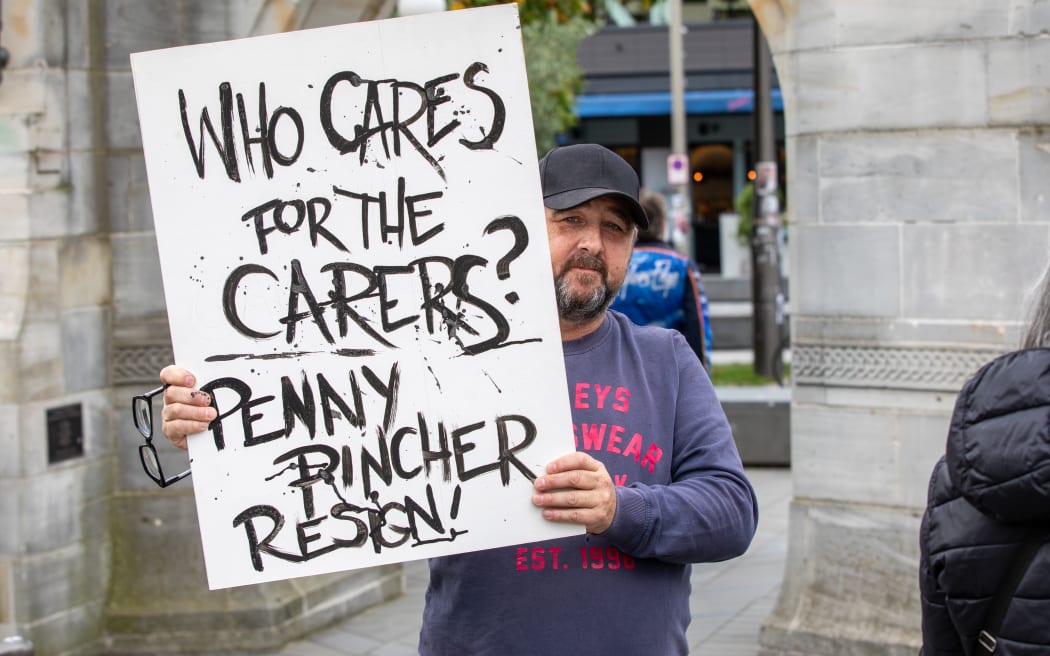


atest comment